Introspective Market Research, a premier market intelligence firm specializing in healthcare and diagnostics, today announced the publication of its comprehensive analysis on the Global Liver Function Test (LFT) Market. LFTs are essential blood tests that analyze enzymes, proteins, and bilirubin levels to assess liver health, diagnose liver disorders, evaluate the extent of damage, and monitor treatment efficacy.
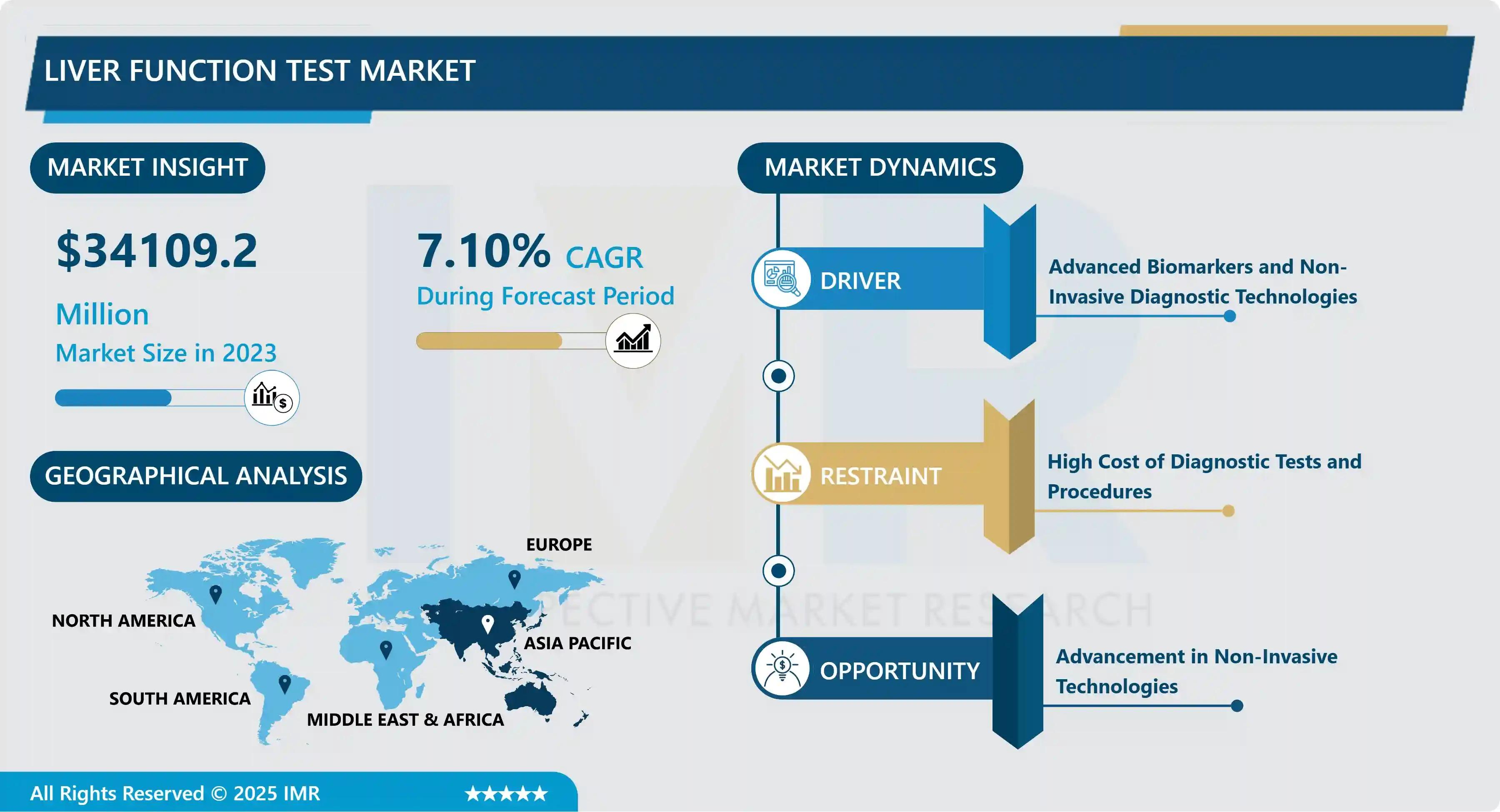
The Global Liver Function Test Market size was valued at USD 34.1 Billion in 2024 and is projected to reach USD 63.2 Billion by 2032, exhibiting a healthy Compound Annual Growth Rate (CAGR) of 7.1% over the 2024–2032 forecast period. This robust growth is primarily driven by the increasing global prevalence of liver diseases such as Hepatitis B and C, cirrhosis, and the rapidly growing epidemic of Non-Alcoholic Fatty Liver Disease (NAFLD) and its more severe form, NASH (Non-Alcoholic Steatohepatitis).
Quick Insights: Global Liver Function Test Market (2024–2032)
| Metric | Insight |
| 2024 Market Valuation | USD 34.1 Billion |
| Projected 2032 Valuation | USD 63.2 Billion |
| CAGR (2024-2032) | 7.1% |
| Dominant Test Type | Alanine Aminotransferase (ALT) Test (Core indicator of hepatocellular injury) |
| Leading End-User | Diagnostic Laboratories (High-throughput testing and sophisticated equipment) |
| Largest Regional Market | Asia Pacific (High incidence of viral hepatitis and NAFLD) |
| Key Market Driver | Rising Prevalence of Chronic Liver Diseases |
| Key Opportunity | Advancement in Non-Invasive Technologies |
Segmentation Spotlight: ALT Test Remains the Core Indicator
The analysis of the LFT market segmentation reveals the foundational nature of traditional enzyme tests, even as new technologies emerge:
- By Product: The Alanine Aminotransferase (ALT) Test segment is expected to dominate the market. ALT is highly specific to the liver, and elevated levels are a primary diagnostic marker for liver damage, including inflammation and injury. Its widespread adoption in routine health screenings, simplicity, and cost-effectiveness ensure its continued dominance across clinical settings.
- By End-User: Diagnostic Laboratories held the largest market share in 2023. These high-volume, centralized facilities are best equipped with the automated systems and cutting-edge technology required to perform LFTs with high precision, speed, and reliability. The shift toward preventative health and routine health check-ups further drives traffic to these specialized labs.
How is the Market Shifting to Non-Invasive Diagnostics and Advanced Biomarkers?
The primary opportunity and prevailing trend in the LFT market is the Advancement in Non-Invasive Technologies and the development of Advanced Biomarkers. Traditional LFTs offer valuable information but are limited in distinguishing the exact cause or severity of liver damage. The market is witnessing a strong shift away from invasive liver biopsies toward non-invasive alternatives, which include:
- Advanced Imaging: Such as transient elastography (FibroScan) to assess liver stiffness.
- Biomarker Assays: New, enhanced blood tests that measure markers of liver fibrosis, steatosis, and inflammation (e.g., FIB-4 score, Enhanced Liver Fibrosis (ELF) test).
These non-invasive methods enhance patient comfort, increase adherence to screening protocols, and allow for more frequent, accurate monitoring of chronic diseases like NASH, ultimately leading to improved patient outcomes and reduced long-term healthcare costs.
Expert Insight: The Need for Standardized Global Screening
“While the demand for LFTs is escalating, particularly due to lifestyle-driven diseases like NAFLD, the challenge lies in standardizing the diagnostic pathway,” comments Dr. Maya Chatterjee, Principal Consultant at Precedence Research. “The sheer volume of testing creates cost pressures for healthcare systems globally. We need to focus on integrating advanced, non-invasive LFT panels into primary care settings and ensure that regulatory bodies around the world establish clear, consistent guidelines for interpreting these sophisticated advanced biomarkers. Without global standardization and favorable reimbursement policies, especially for the high-volume ALT testing, preventative liver health will remain segmented and inefficient.”
Regional Dynamics and Corporate Strategy
The Asia Pacific region is expected to dominate the market over the forecast period. This leadership is driven by the alarmingly high incidence of viral hepatitis (B and C) and the rapidly rising prevalence of metabolic disorders leading to NAFLD, particularly in countries like China and India. Increased government investment in healthcare infrastructure and public awareness campaigns promoting early disease detection further accelerate market growth.
In contrast, North America and Europe, while mature, remain strong markets, focused on integrating sophisticated, automated LFT analyzers and non-invasive diagnostic equipment into clinical practice.
Corporate Breakthroughs: Key players such as Roche Diagnostics and Abbott Laboratories are focusing on developing high-throughput, automated immunoassay systems that can run a comprehensive panel of LFTs and advanced biomarkers simultaneously, increasing lab efficiency. Companies like Siemens Healthineers are pushing the envelope with compact, point-of-care LFT solutions, enabling rapid results outside of centralized laboratories.
Challenges: Volume and Data Interpretation
The high volume of routine testing creates a fundamental challenge regarding assay consistency and cost-effectiveness. While the cost per test is generally low, the sheer frequency of LFTs globally pressures reimbursement rates. Another challenge lies in data interpretation: the increasing use of advanced panels means clinicians must effectively interpret complex patterns across multiple biomarkers, necessitating better training and integration with electronic health records (EHRs) for personalized medicine.
Case Study: Point-of-Care LFT Integration
A network of community health clinics in a large US metropolitan area integrated a new rapid, point-of-care (POC) LFT analyzer. Before the POC system, patients had to wait 24-48 hours for lab results, leading to delays in follow-up. With the POC system, patients received LFT results within minutes during their initial visit. This change facilitated immediate discussion of elevated enzyme levels, expedited referrals for specialized treatment (like FibroScan), and improved patient compliance with diagnostic recommendations by over 50%, demonstrating the clear clinical utility of faster, decentralized testing.
Call to Action
Diagnose Tomorrow's Health: Download the Full Liver Function Test Market Report
About Introspective Market Research
Introspective Market Research (IMR) is a trusted provider of comprehensive market intelligence, offering in-depth insights into global industry trends, competitive landscapes, and growth opportunities. Our reports empower businesses to make informed, strategic decisions that accelerate growth and maximize value across diverse sectors.
Contact: Introspective Market Research Phone: +91-74101-03736
Email: sales@introspectivemarketresearch.com
Website: https://introspectivemarketresearch.com/